The Bulk lyophilization services play a vital role in the pharmaceutical and biotechnology industries by enabling the stabilization of temperature sensitive drugs. Freeze drying technology helps convert liquid biologics into stable solid forms, ensuring longer shelf life and easier transportation. As the complexity of therapeutics increases, the need for reliable preservation solutions continues to grow across global healthcare systems.
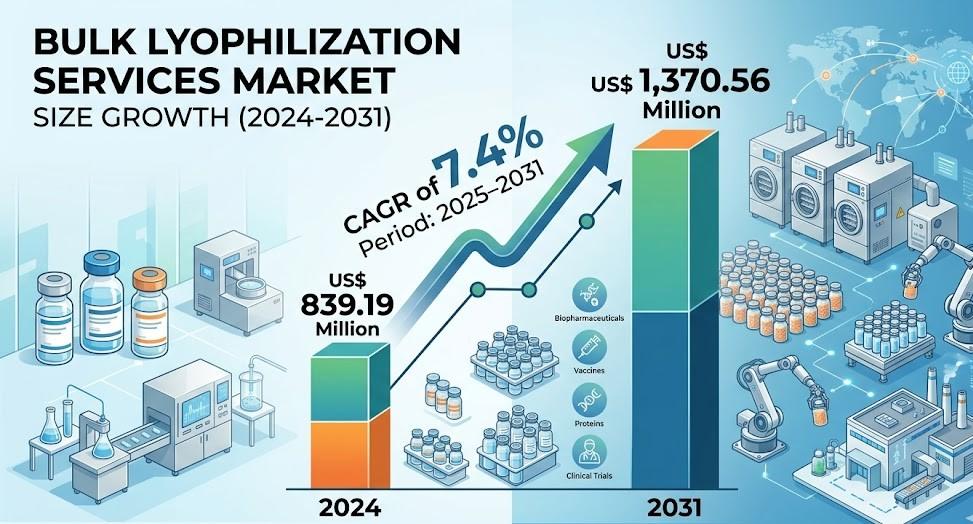
The Bulk Lyophilization Services Market Analysis reflects a strong upward trajectory, with the Bulk lyophilization Services Market valued at US$ 839.19 million in 2024 and projected to reach US$ 1,370.56 million by 2031. This expansion represents a compound annual growth rate of 7.4% during the forecast period from 2025 to 2031. Growth is largely driven by increasing biologics production, rising vaccine demand, and the growing preference for outsourcing manufacturing processes to specialized service providers.
Increasing Demand for Biologics and Vaccines
The surge in biologics development is a primary factor fueling demand for bulk lyophilization services. Products such as monoclonal antibodies, vaccines, and gene therapies are highly sensitive to environmental conditions and require advanced preservation methods. Lyophilization enhances their stability by removing moisture, allowing these therapies to maintain efficacy over extended periods.
In addition, the global expansion of vaccination programs has created a strong need for stable formulations that can be transported across regions with limited cold chain infrastructure. Lyophilized vaccines offer improved storage flexibility, making them suitable for distribution in diverse climatic conditions. This trend continues to support the adoption of bulk freeze drying technologies.
Download Sample PDF @ https://www.theinsightpartners.com/sample/TIPRE00041488
Growing Shift Toward Outsourcing
Pharmaceutical companies are increasingly outsourcing lyophilization processes to contract development and manufacturing organizations. Setting up in house lyophilization facilities involves significant capital investment, strict regulatory requirements, and operational complexity. Outsourcing allows companies to reduce costs while accessing advanced technologies and specialized expertise.
This approach also provides flexibility in scaling production based on demand. As biologics pipelines expand, companies prefer to collaborate with experienced service providers who can handle large volume production efficiently. The rise in strategic partnerships between pharmaceutical firms and contract manufacturers highlights the importance of outsourcing in the current landscape.
Technological Advancements and Process Optimization
Technological innovation is transforming the bulk lyophilization services industry. Advanced techniques such as controlled ice nucleation, process analytical technology, and quality by design are improving process efficiency and product consistency. These innovations enable better control over freezing and drying stages, ensuring uniform quality across large batches.
Automation is another key development, with the integration of robotics and digital monitoring systems reducing human intervention and minimizing contamination risks. Real time analytics powered by artificial intelligence allow operators to monitor critical parameters such as temperature and pressure, leading to optimized cycle times and improved productivity. These advancements are making lyophilization more scalable and cost effective.
Segmentation Insights and Service Expansion
The market is segmented by lyophilization format, scale, service type, and end user, providing a comprehensive view of industry dynamics. Bulk tray lyophilization holds the largest share due to its efficiency in handling large volumes. Other formats such as drum lyophilization, shelf freeze drying, and tunnel based systems cater to specialized requirements.
Commercial and industrial scale operations are expected to witness the fastest growth, driven by increasing demand for large scale production. In terms of service type, full scale commercial bulk lyophilization dominates, reflecting the preference for end to end service offerings.
Pharmaceutical and biotechnology companies are the leading end users, supported by their extensive research pipelines and growing production needs. Research institutions also contribute to demand, particularly in early stage development and clinical trials.
Regional Trends and Competitive Landscape
North America accounts for the largest share of the global market, supported by advanced healthcare infrastructure, strong research activity, and the presence of numerous contract manufacturing organizations. The region’s strict regulatory standards further encourage the adoption of high quality lyophilization processes.
Asia Pacific is expected to register the fastest growth, with a projected CAGR of 7.8% during the forecast period. This growth is driven by expanding biopharmaceutical manufacturing capacity, government support, and cost advantages. Countries such as India and China are becoming prominent hubs for outsourced pharmaceutical production.
Europe also holds a significant share due to early adoption of advanced technologies and a strong regulatory environment. Meanwhile, regions such as Latin America and the Middle East and Africa are gradually emerging as potential growth areas due to improving healthcare infrastructure and increasing demand for vaccines.
The competitive landscape is characterized by the presence of both global and regional players focusing on innovation and expansion.
Key Players:
- ATTWILL Medical Solutions
- PCI Pharma Services
- Lyophilization Technology, Inc.
- OFD Life Sciences
- Symbiosis Pharmaceutical Services Ltd
- Quality BioResources
- Affinity Life Sciences, Inc.
- Alcami Corporation
- Hudson Valley Lyomac
- Catachem Inc.
Future Outlook
The future of the bulk lyophilization services market is promising, driven by the continued growth of biologics and advanced therapeutics. Increasing reliance on outsourcing, combined with ongoing technological advancements, is expected to enhance efficiency and scalability. Emerging applications such as cell and gene therapies will create new opportunities for specialized lyophilization solutions. Additionally, expansion into emerging markets will further support global growth. As the pharmaceutical industry evolves, bulk lyophilization will remain a critical technology for ensuring the stability and accessibility of modern therapeutics.
Related Reports @
Bupivacaine Liposome Injectable Suspension Market Drivers, Trends, and Forecasts by 2031
Sutherlandia Extract Market Research Report by Size Share Analysis 2031
About Us -
The Insight Partners provides comprehensive syndicated and tailored market research services in the healthcare, technology, and industrial domains. Renowned for delivering strategic intelligence and practical insights, the firm empowers businesses to remain competitive in ever-evolving global markets.
Contact Us:
Ankit Mathur | The Insight Partners
E-mail: ankit.mathur@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in : Korean| German| Japanese| French| Chinese| Italian| Spanish